The problem with pills
The stomach destroys most of what you swallow.
Gastric acid sits at a pH of 1.5 to 3.5. Most supplement compounds — fat-soluble vitamins, botanical extracts, amino acids — are partially or fully degraded before they reach the small intestine, the site where absorption actually occurs.
Compressed tablets and standard capsules dissolve indiscriminately. There is no mechanism to protect a compound through the stomach or to time its release for the intestinal region where it is most efficiently absorbed.
Bioavailability — the fraction of a dose that reaches systemic circulation — varies between 5% and 95% depending on the compound and delivery format. A poorly delivered formula is not a formula. It is an expensive exercise in hope.
Microgranule technology
Pharmaceutical-grade delivery.
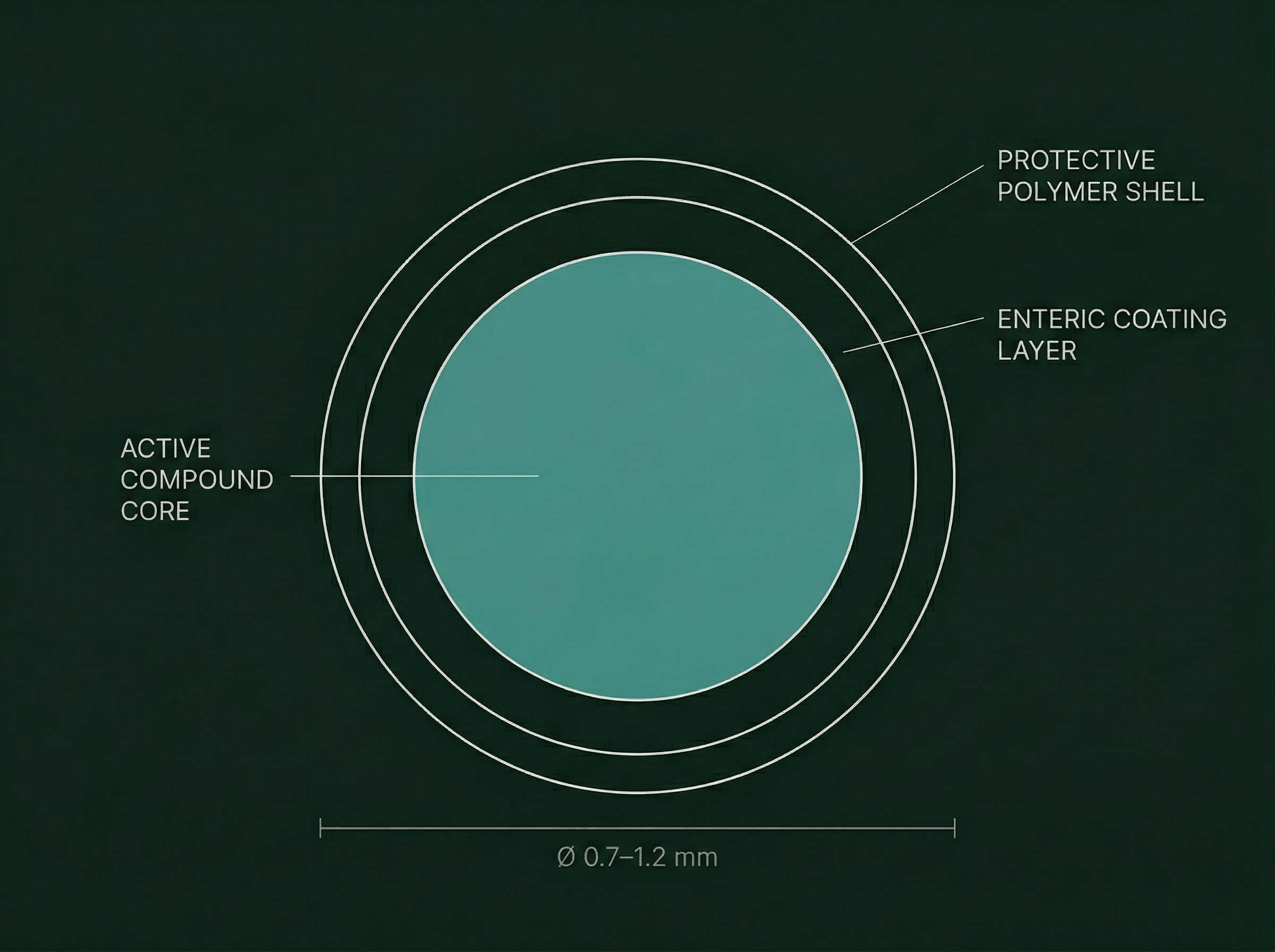
Each PARTICULAR formula is blended as spherical microgranules of 0.7–1.2mm, produced by pharmaceutical extrusion-spheronization — a process with a 30-year history in pharmaceutical medicine and a proven safety record across thousands of licensed drug formulations.
Each granule is individually coated to control exactly where in the digestive tract it dissolves. The coating material, its thickness, and its composition are calibrated per ingredient — not per formula.
The process is patented. The technology is validated in peer-reviewed literature. The facility is GMP-certified. This is not standard supplement manufacturing — it is the same microgranule technology used in pharmaceutical production, applied here to personalised nutrition.
Delivery in action
Three stages.
Optimum absorption.
From dry particle to intestinal absorption — the enteric coating protects each compound through the stomach's acid environment, then dissolves precisely at the point of highest absorption efficiency.
Four mechanisms.
One monthly blend.
Gastric protection
The enteric coating resists the acidic environment of the stomach. Active compounds reach the intestine intact — where absorption is both possible and efficient.
Controlled release
Coating thickness determines release rate. Oil-soluble compounds are released slowly into the upper intestine. Water-soluble compounds release as the coating dissolves. Each ingredient behaves exactly as intended.
Dose uniformity
Each microgranule contains the same concentration of active ingredient. When 300mg of Ashwagandha is specified, 300mg of active compound is delivered — not an approximation.
Independent release profiles
Compounds that would compete for absorption in a standard capsule are separated by coating. Each ingredient completes its own release cycle without interference from others.
Ingredient selection
The form matters as much as the dose.
KSM-66® Ashwagandha
Root extract only. No leaf content. ≥5% withanolides standardised. The most clinically studied ashwagandha extract — over 20 randomised controlled trials.
MenaQ7® Vitamin K2 MK-7
The only form of K2 with a ~72-hour half-life in circulation. Derived from natto. Patented extraction process. Clinically validated for bone and cardiovascular support.
Vegan Cholecalciferol (D3 from lichen)
Cholecalciferol — the same form produced by sunlight exposure — sourced from lichen rather than lanolin. Identical bioavailability. Suitable for vegans.
5-MTHF Folate
The biologically active form of folate. Bypasses the MTHFR conversion step that affects approximately 40% of the population — making it more readily utilised for those with varying folate metabolism.
Methylcobalamin (B12)
The neurologically active form. Directly usable by the body without hepatic conversion — well-suited to those with varying B12 metabolic requirements.
Quality & provenance
Every batch. Every ingredient. Verified.
Manufactured in a GMP-certified facility in Germany. Every production run subject to release testing before dispatch.
The microgranule process is protected by three independent patents covering the manufacturing method, coating application, and particle geometry. The technology is not replicated by the generic supplement industry.
Peer-reviewed validation in pharmaceutical journals — not nutritional supplement trade press — confirms delivery efficacy across multiple compound classes.
